NCERT Solutions for Class 9 Science (Chemistry) Chapter 2 Is Matter Around Us Pure provides detailed answers for all in-text and exercise Questions. These solutions contain an in-depth explanation of each topic involved in the chapter. Students studying in class 9 can access these solutions for free in PDF format.
All these solutions are prepared by expert teachers and updated for the current academic session. NCERT Solutions for Class 9 Science (Chemistry) Chapter 2 Is Matter Around Us Pure help students to understand the fundamental concepts given in class 9 Science textbook. We have prepared the answers to all the questions in an easy and well-structured manner. It helps students to grasp the chapter easily.
Class 9 Science Is Matter Around Us Pure Intext Questions and Answers
PAGE NO. 15
Questions 1: what do mean by a pure substance?
Answer: A pure substance is a material that has a constant composition and consistent properties throughout the sample. It can be either an element, which is made up of only one kind of atom, or a compound, which is made up of two or more types of atoms chemically bonded together in a fixed ratio.
Question 2: List the points of differences between homogeneous and heterogeneous mixtures.
Answer:
| Homogeneous mixture | Heterogeneous mixture |
| A homogeneous mixture is a mixture having a uniform composition throughout the mixture. | A heterogeneous mixture is a mixture having a non-uniform composition throughout the mixture. |
| Has a uniform composition | Irregular composition |
| No apparent boundaries of division | Noticeable boundaries of division. |
| For example: salt in water, sugar in water, copper sulphate in water | For example: sodium chloride and iron filings, salt and sulphur, oil and water |
PAGE NO. 18
Question 1: Differentiate between homogeneous and heterogeneous mixtures with examples.
Answer: A homogeneous mixture is a mixture having a uniform composition throughout the mixture. For example, mixtures of salt in water, sugar in water, copper sulphate in water, iodine in alcohol, alloy, and air have uniform compositions throughout the mixtures.
On the other hand, a heterogeneous mixture is a mixture having a non-uniform composition throughout the mixture. For example: the composition of mixtures of sodium chloride and iron filings, salt and sulphur, oil and water, chalk powder in water, wheat flour in water, milk and water are not uniform throughout the mixtures.
Question 2: How are sol, solution and suspension different from each other?
Answer:
| Sol | Solution | Suspension |
| Sol is a heterogeneous mixture. | Solution is a homogeneous mixture. | Suspensions are heterogeneous mixtures. |
| Size of particles lies between 10-7 – 10-5 cm | Size of particles are less than 1nm | Size of particles are more than 100nm |
| Tyndall effect is observed | Tyndall effect is not observed | Tyndall effect is observed |
| Solute particles cannot be seen with the naked eye | Solute particles cannot be seen with the naked eye | Solute particles are visible to the naked eye |
| Diffuses very slowly | Diffuses rapidly | Do not diffuse |
| For example. Milk, blood, smoke | For example. Salt solution, Sugar solution | For example. Sand in water, dusty air |
Question 3: To make a saturated solution, 36 g of sodium chloride is dissolved in 100 g of water at 293 K. Find its concentration at this temperature.
Answer: Mass of solute (sodium chloride) = 36 g (Given)
Mass of solvent (water) = 100 g (Given)
Then, mass of solution = Mass of solute + Mass of solvent
= (36 + 100) g
= 136 g
Therefore, concentration (mass by mass percentage) of the solution
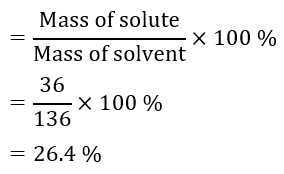
PAGE NO. 19
1. Classify the following as physical or chemical changes:
- Cutting of trees
- Melting of butter in a pan
- Rusting of almirah
- Boiling of water to form steam
- Passing of electric current through water and water breaking into hydrogen and oxygen gases.
- Dissolving common salt in water
- Making a fruit salad with raw fruits, and
- Burning of paper and wood
Answer: The following is the classification into physical and chemical change
| Physical change | Chemical change |
| Cutting the trees | Rusting of almirah |
| Boiling of water to form steam | Passing of electric current through water, |
| Melting of butter in a pan | water breaking into hydrogen and oxygen gases |
| Making a fruit salad with raw fruits | |
| Dissolving common salt in water |
Question 2: Try segregating the things around you as pure substances or mixtures.
Answer: Pure substance: Water, salt, sugar etc.
Mixture: Salt water, soil, wood, air, cold drink, rubber, sponge, fog, milk, butter, clothes, food.
Class 9 Science Matter in Our Surroundings Exercise Questions
Question 1: Which separation techniques will you apply for the separation of the following?
(a) Sodium chloride from its solution in water.
(b) Ammonium chloride from a mixture containing sodium chloride and ammonium chloride.
(c) Small pieces of metal in the engine oil of a car.
(d) Different pigments from an extract of flower petals.
(e) Butter from curd.
(f) Oil from water.
(g) Tea leaves from tea.
(h) Iron pins from sand.
(i) Wheat grains from husk.
(j) Fine mud particles suspended in water.
Answer:
(a) Crystallization or Evaporation.
(b) Sublimation.
(c) Centrifugation or Sedimentation.
(d) Chromatography.
(e) Centrifugation.
(f) Separating funnel.
(g) Hand-picking.
(h) Magnetic separation.
(i) Winnowing.
(j) Centrifugation.
Question 2: Write the steps you would use for making tea. Use the words – solution, solvent, solute, dissolve, soluble, insoluble, filtrate and residue.
Answer: Take the solvent, water, in a kettle. Heat it. When the solvent boils, add the solute, milk. Milk and water forms a solution. Then pour some tea leaves over a sieve. Pour slowly hot solution of milk over tea leaves. Colour of tea leaves goes into solution as filtrate. The remaining tea leaves being insoluble remains as residue. Add requisite sugar which dissolves and the tea is ready.
Question 3. Pragya tested the solubility of three different substances at different temperatures and collected the data as given below (results are given in the following table, as grams of a substance dissolved in 100 grams of water to form a saturated solution).
| Temperature in K | |||||
| Substance Dissolved | 283 | 293 | 313 | 333 | 353 |
| Potassium nitrate | 21 | 32 | 62 | 106 | 167 |
| Sodium chloride | 36 | 36 | 36 | 37 | 37 |
| Potassium chloride | 35 | 35 | 40 | 46 | 54 |
| Ammonium chloride | 24 | 37 | 41 | 55 | 66 |
(a) What mass of potassium nitrate would be needed to produce a saturated solution of potassium nitrate in 50 grams of water at 313 K?
(b) Pragya makes a saturated solution of potassium chloride in water at 353 K and leaves the solution to cool at room temperature. What would she observe as the solution cools? Explain.
(c) Find the solubility of each salt at 293 K. Which salt has the highest solubility at this temperature?
(d) What is the effect of change of temperature on the solubility of a salt?
Answer: (a) At 313 K,
Potassium nitrate for saturated solution of 100 grams of water = 62 g
Potassium nitrate for saturated solution of 50 grams of water = 31 g
(b) Some amount of dissolved Potassium Chloride will reappear as undissolved solid as solubility of solute decreases with the decrease of temperature.
(c) Solubility of each salt at 393 K are as follows:
- Potassium nitrate – 32 g
- Sodium chloride – 36 g
- Potassium chloride – 35 g
- Ammonium chloride – 37 g
Ammonium chloride salt has the highest solubility at this temperature.
(d) Solubility of salt increases with the increase in temperature.
Question 4: Explain the following giving examples.
(a) Saturated solution,
(b) Pure substance,
(c) Colloid,
(d) Suspension.
Answer: (a) Saturated Solution: A solution in which no more of the solid (solute) can be dissolved at a given temperature is called a saturated solution. Suppose 50 gm of a solute is the maximum amount that can be dissolved in 100 gm water at 298 K. Then 150 gm of solution so obtained is the saturated solution at 298 K.
(b) Pure Substance: A pure substance consists of a single of matter or particles and cannot be separated into other kind of matter by any physical process. Pure substances always have the same colour, taste and texture at a given temperature and pressure. For example, pure water is always colourless, odourless and tasteless and boils at 373 K at normal atmospheric pressure.
(c) Colloid: Colloids are heterogeneous mixtures the particle size is too small to be seen with a naked eye, but it is big enough to scatter light. The particles are called the dispersed phase and the medium in which they are distributed is called the dispersion medium. Colloids are useful in industry and daily life. A colloid has the following characteristics:
- It is a heterogeneous mixture.
- The size of particles of a colloid lies between 1 – 100 nm and cannot be seen by naked eyes.
- The particles of colloid can scatter a beam of light passing through it and make the path visible.
- The particles of colloid cannot be separated from the mixture by filtration. The process of separation of colloidal particles is known as ‘centrifugation’.
- They do not settle down when left undisturbed. In other words, colloids are quite stable e.g. smoke, milk, fog, cloud etc.
(d) Suspension: It is a heterogeneous mixture that comprises of solute particles that are insoluble but are suspended in the medium. These particles that are suspended are not microscopic but visible to bare eyes and are large enough (usually larger than a micrometer) to undergo sedimentation.
Question 5. Classify each of the following as a homogeneous or heterogeneous mixture: soda water, wood, air. soil, vinegar, filtered tea.
Answer: Homogeneous: Soda water, vinegar, filtered tea.
Heterogeneous: Wood, air, soil.
Question 6. How would, you confirm that a colourless liquid given to you is pure water?
Answer: By finding the boiling point of a given colourless liquid. If the liquid boils at 100°C at atmospheric pressure, then it is pure water. This is because pure substances have fixed melting and boiling point.
Question 7. Which of the following materials fall in the category of a “pure substance”?
(a) Ice
(b) Milk
(c) Iron
(d) Hydrochloric acid
(e) Calcium oxide
(f) Mercury
(g) Back
(h) Wood
(i) Air.
Answer: Pure substances are: Ice, iron, hydrochloric acid, calcium oxide and mercury.
Question 8. Identify the solutions among the following mixtures.
(a) Soil
(b) Seawater
(c) Air
(d) Coal
(e) Soda water.
Answer: Solutions are: Sea water soda water and air.
Question 9. Which of the following will show “Tyndall effect”?
(a) Salt solution
(b) Milk
(c) Copper sulphate solution
(d) Starch solution.
Answer: Milk and starch solution.
Question 10. Classify the following into elements, compounds and mixtures.
(a) Sodium
(b) Soil
(c) Sugar solution
(d) Silver
(e) Calcium carbonate
(f) Tin
(g) Silicon
(h) Coal
(i) Air
(j) Soap
(k) Methane
(l) Carbon dioxide
(m) Blood
Answer:
| Elements | Compounds | Mixture |
| Sodium | Calcium carbonate | Soil |
| Silver | Carbon dioxide | Sugar solution |
| Tin | Methane | Coal |
| Silicon | Air | |
| Blood | ||
| Soap |
Question 11. Which of the following are chemical changes?
(a) Growth of a plant
(b) Rusting of iron
(c) Mixing of iron filings and sand
(d) Cooking of food
(e) Digestion of food
(f) Freezing of water
(g) Burning of a candle.
Answer: Chemical changes are:
(a) Growth of a plant
(b) Rusting of iron
(c) Cooking of food
(d) Digestion of food
(e) Burning of a candle